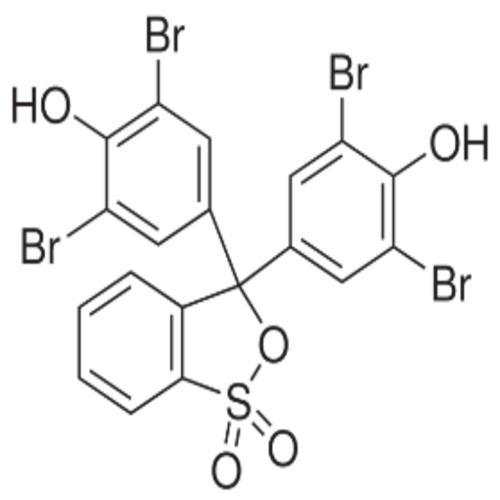
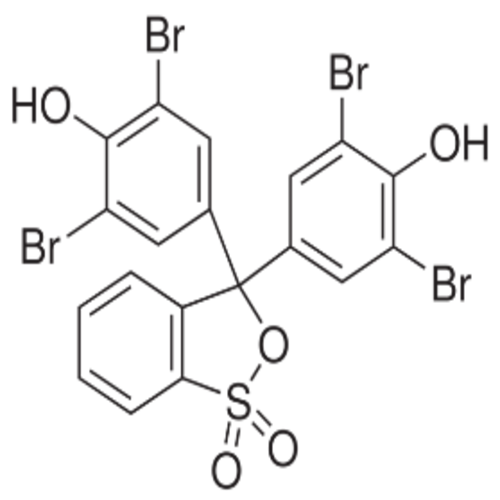
Bromophenol Blue
Price 5000 INR/ Container
Bromophenol Blue Specification
- Molecular Formula
- C19H10Br4O5S
- CAS No
- 115-39-9
Bromophenol Blue Trade Information
- Minimum Order Quantity
- 1 Container
- Payment Terms
- Cheque
- Supply Ability
- 25 Containers Per Week
- Delivery Time
- 15 Days
- Packaging Details
- 1-25kg in drum
- Main Domestic Market
- All India
About Bromophenol Blue
Chemical Name
Phenol, 4,4'-(3H-2,1-benzoxathiol-3-ylidene)bis[2,6-dibromo-, S,S-dioxide
InChI Key
UDSAIICHUKSCKT-UHFFFAOYSA-N
Solubility
Soluble in acetic acid.Soluble in water (about 0.4 g/100 mL); more soluble in methyl and ethyl alcohol, and in benzene. Freely soluble in NaOH solutions with the formation of a water-soluble sodium salt.In water, 3 mg/mL
Product Description:
Bromophenol blue is an acid-base indicator since its useful range lies between pH 3.0 and 4.6. It changes from yellow at pH 3.0 to blue at pH 4.6; this reaction is reversible. Bromophenol blue is structurally related to phenolphthalein (a popular indicator). Bromophenol blue is also used as a color marker to monitor the process of agarose gel electrophoresis and polyacrylamide gel electrophoresis
Applications:
Chemicals and substances that impart color including soluble dyes and insoluble pigments. They are used in INKS; PAINTS; and as INDICATORS AND REAGENTS.Concentrations of bromophenol blue (I) in plasma, urine, and bile were determined spectrophotometrically after intravenous bolus injections and infusions in rats. The plasma concentrations were found to decrease mono exponentially after all doses except the highest, where the decrease was biexponential. Bromophenol blue (BPB) is the most widely used stain in gel electrophoresis for tracing the migration of samples on electrophoretic gels. If the gel is staind with 1 - 5 mg bromophenol blue, this may be detected much easier and earlier. In case that the stain will interfere with other applications (e. g. silver staining), a pre-run of the gel will remove bromophenol blue. It does not influence the migration of the samples. Add the solid stain before adding the catalyst.
Product details
|
M.F. |
C19H10Br4O5S |
|
Country of Origin |
Made in India |
|
CAS Number |
115-39-9 |
|
Chemical Formula |
C19H10Br4O5S |
|
Packaging Size |
1- 25 kgs |
|
Packaging Type |
drum |
- Product : Bromophenol Blue
- C.A.S No. : 115-39-9
- M.F. : C19H10Br4O5S
- M.W. : 669.99gm/mole
Specification
|
Dye Content (by Spectrophotometry) |
>95% |
|
Transition Range |
pH 3.0-4.6 |
|
|
Greenish Yellow-Blue |
|
Solubility 0.1% (95% EtOH) |
Clear solution |
|
Absorption maximum (pH 3.0) 1max |
434-439nm |
|
Absorption maximum (pH 4.6) 2max |
590-593nm |
|
Absorptivity (A1%/1cm at pH 3.0 at 1max) |
350-385 |
|
Absorptivity (A1%/1cm at pH 4.6 at ?2max) |
940-1000 |
|
Loss On Drying (110 C) |
<1% |

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Lab Reagents Category
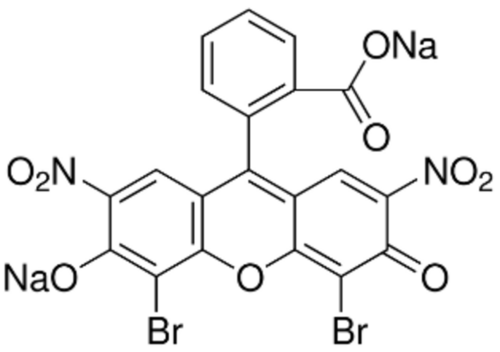
Phloxine B Chemical
Price 19000 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 18472872
Molecular Formula : C20H2Br4Cl4O5
Molecular Weight : 829.74 g/mol
Eosin Blue Chemical
Price 4000 INR / Container
Minimum Order Quantity : 1 Container
CAS No : 548243
Molecular Formula : C20H6Br2N2Na2O9
Molecular Weight : 624.09gm/mole GSM (gm/2)
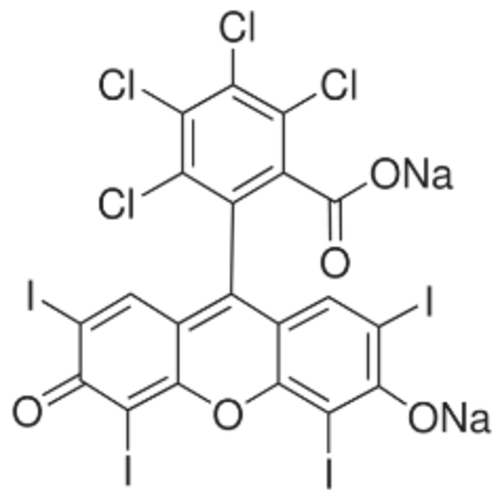
Rose Bengal 90% Chemical
Price 10500 INR / Container
Minimum Order Quantity : 1 Container
CAS No : 632699
Molecular Formula : C20H2O5Cl4I4Na2
Molecular Weight : 1017.65gm/mole GSM (gm/2)
Bromothymol Blue
Price 8000 INR / Container
Minimum Order Quantity : 1 Container
CAS No : 76595
Molecular Formula : C27H28Br2O5S
 |
KARAN LABORATES
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry